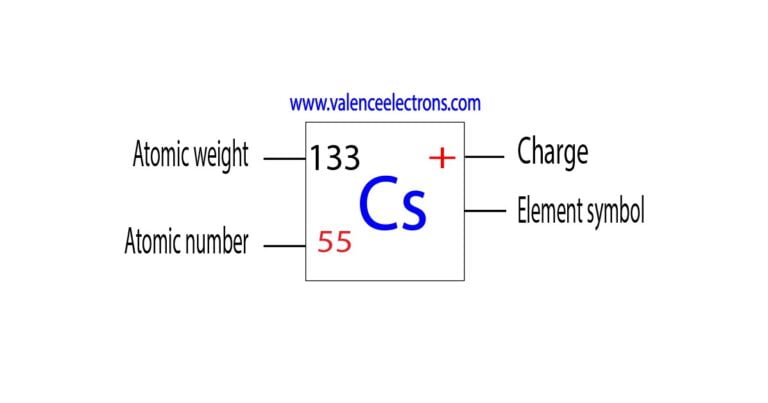
Cesium atoms condense on the electrodes, changing their effective work function in a. We noted that such irregularities were due to the radius of Ca 2+ 2+ in the AMBER force field is the vdW radius for the atomic Lennard-Jones potential of calcium, while the ionic radius of Ca 2+ should be smaller. Cesium ions can cancel the negative interelectrode space charge. Caesium ions have a 1+ charge and iodide ions have a 1- charge. It is a soft, silvery-golden alkali metal with a melting point of 28.5 C (83.3 F). This is dependent on the magnitude of the ionic charges. Trending Questions How may the Earth's position in space be related to the cycle of extinctions earth? Why could ancient observers only see five of the planets? What causes 1 revolution of the Earth? How far can a candle be seen in light? How is gravity both a friend and enemy of a star? What the Europeans learned from the Arabs? Which stellar properties has the greatest range in values? Why do astromers study stars today? Do astronomers get insurance? 1.Pluto is how far away from earth 2.How far away from the sun 3. Comparison between the iRESP charges of the Ca 2+ ion from the electrostatic potential using the vdW radii of Ca 2+ vdW. Caesium is a chemical element with the symbol Cs and atomic number 55. Distributions of bare cesium cluster ions also indicate enhanced stability of Cs3 and Cs5, in agreement with theoretical predictions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
